 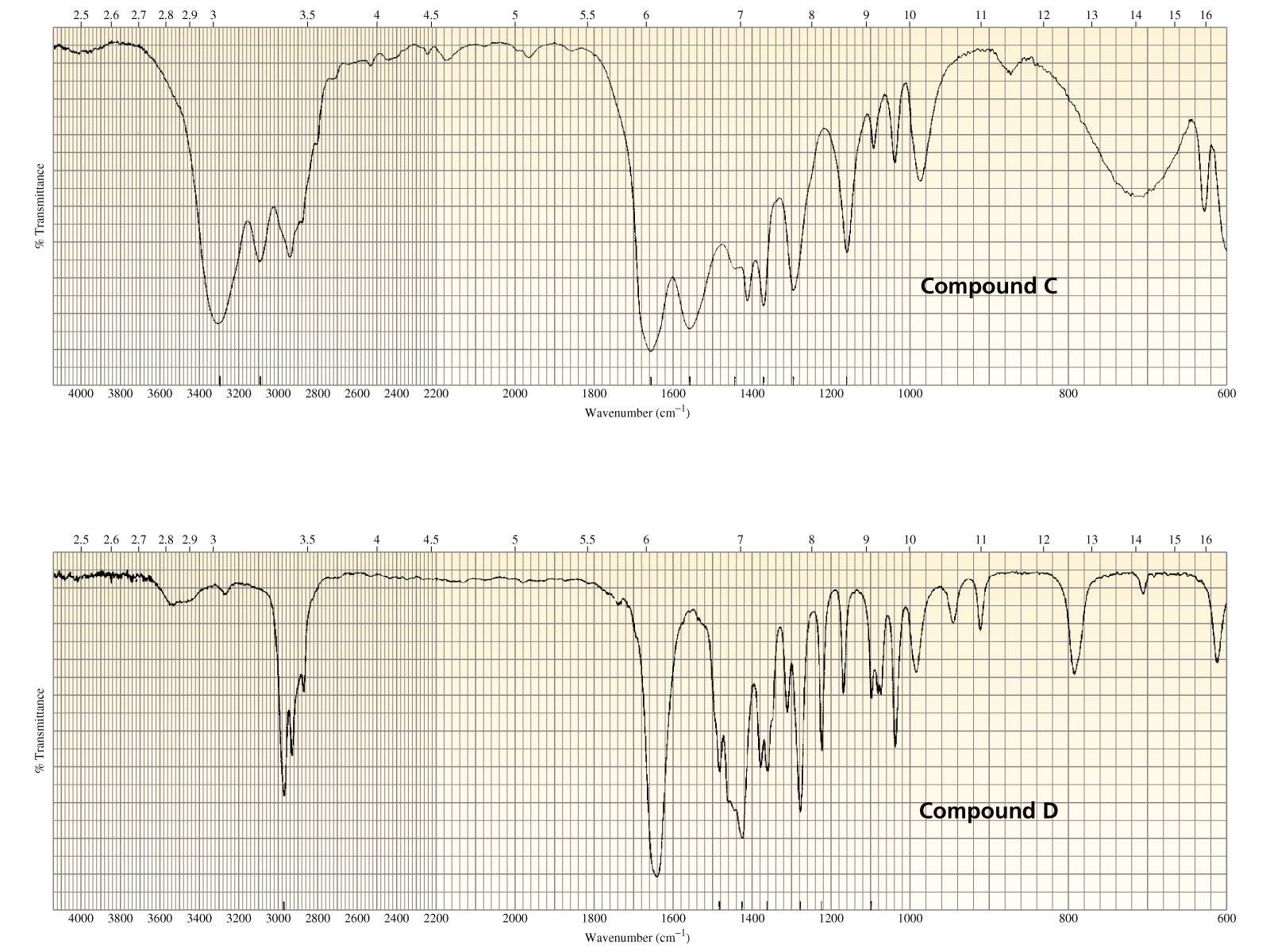
With this conclusive proof our formula, as we have written it, must be correct and our ideas in regard to the oxidation of compounds containing hydrogen linked to carbon are probably correct also. The conclusive proof that in acetone there are two methyl groups present is in the synthesis of acetone from acetic acid and acetyl chloride, reactions which we shall soon study. Although this may function purely as a base to neutralize the hydrochloric acid which is formed in the reaction, bases such as dimethylaminopyridine can also activate the carboxyl group via the formation of the intermediate shown in Scheme 3.66. The reaction is often facilitated by the presence of a tertiary amine catalyst. Īcyl halides such as acetyl chloride react with water to regenerate the starting acid and they react with alcohols to yield esters. Similarly, a-acetoxyrhenacyclobutadiene complexes, generated in situ by the reaction of acetyl chloride with rhenacyclobutanone 14, undergo substitution upon reaction with alcohols (Equation 3), giving a-alkoxyrhenacyclo-butadiene complexes 15.  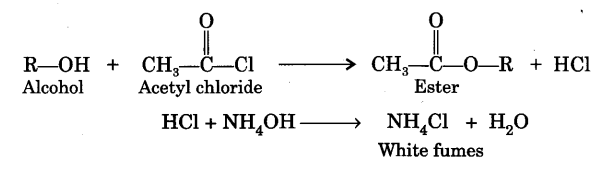
Acetyl chloride will add across unsaturated bonds in the presence of suitable catalysts 10 give halogenated ketunes. Acetyl chloride reaction with alcohols Reaction with alcohols gives the corresponding acetate esters.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
